CARsgen Therapeutics Announces 2025 Annual Results
Business Highlights
- Cash and cash equivalents were around
RMB1,123 million as ofDecember 31, 2025 . Cash and cash equivalents at the end of 2026 are expected to be not less thanRMB1,000 million . The net loss for the year endedDecember 31, 2025 wasRMB103 million , representing a decrease of approximately 87% compared to the year endedDecember 31, 2024 . In light of operational factors such as the changes in operating cash flow, we expect to have adequate cash into the 2030. - During 2025, CARsgen has received a total of 218 confirmed orders of zevor-cel from its commercialization partner Huadong Medicine. In
December 2025 , zevor-cel has been included inChina's Commercial Health Insurance Innovative Drug Catalogue (2025). -
The Center for Drug Evaluation (CDE) of theNational Medical Products Administration (NMPA) ofChina has accepted the New Drug Application (NDA) and granted Priority Review for satri-cel. The results of satri-cel confirmatory Phase II trial inChina have been simultaneously published inThe Lancet and at the 2025 ASCO Annual Meeting. - Multiple allogeneic CAR-T products are in development, covering treatment areas such as hematologic malignancies, solid tumors, and autoimmune diseases.
- CARsgen independently developed the lentiviral-based CARvivo™ platform for creating in vivo CAR T-cell products.
- CARsgen entered into strategic cooperation agreements with a key platform enterprise in
Jinshan District ,Shanghai to establish an advanced commercial manufacturing base for CAR T-cell products.
Dr. Zonghai Li, Founder, Chairman of the Board, Chief Executive Officer, and Chief Scientific Officer of
Financial Highlights
CARsgen's revenue was around
Cash and cash equivalents were around
Zevor-cel demonstrates rapids sales growth
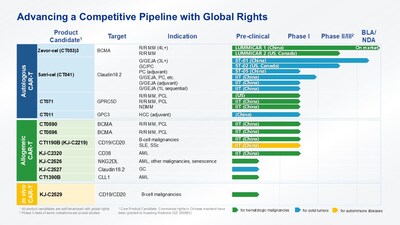
Zevorcabtagene autoleucel (zevor-cel, R&D code: CT053) is an autologous fully human CAR T-cell product against B-cell maturation antigen (BCMA) approved by the NMPA of
CARsgen entered into a collaboration agreement with Huadong Medicine (000963.SZ) for the commercialization of zevor-cel in mainland
The updated long-term follow-up results of Phase I clinical trial of CT053 have been published in Blood Advances. The updated data of Phase II clinical trial, involving 102 patients with a median follow-up of 20 months, were published in Experimental Hematology & Oncology. Zevor-cel demonstrates manageable safety profile while eliciting deep and durable responses in R/R MM patients.
CARsgen entered into a collaboration with Dispatch Bio, to conduct a Phase I trial in
Satri-cel is about to be commercialized in
Satricabtagene autoleucel (satri-cel, R&D code: CT041) is an autologous humanized CAR T-cell product against Claudin18.2. In
The results of satri-cel confirmatory Phase II trial (NCT04581473) in
The research results of the Phase Ib registrational clinical trial of satri-cel for PC adjuvant therapy in
In addition, an investigator-initiated trial (IIT) for satri-cel be used as consolidation treatment following adjuvant therapy in patients with resected G/GEJA (NCT06857786), and an IIT for satri-cel as a sequential therapy following first-line treatment in patients with advanced G/GEJA (NCT07179484) have been conducted in
Multiple allogeneic CAR-T product candidates in development
CARsgen has been advancing differentiated allogeneic CAR T-cell products utilizing the CARsgen's proprietary THANK-uCAR® and THANK-u Plus™ platform. THANK-u Plus™ platform, as an enhanced version of THANK-uCAR®, was developed to address the potential impact of NKG2A expression levels on therapeutic efficacy of the allogeneic CAR T-cells.
CT0596 is a BCMA-targeting allogeneic CAR T-cell product candidate deploying our THANK-u Plus™ technology. The IITs are ongoing in
CT1190B (KJ-C2219) is an allogeneic CAR T-cell product candidate targeting CD19/CD20 deploying our THANK-u Plus™ technology, for hematologic malignancies and autoimmune diseases. The IITs have been initiated for R/R B-NHL, and for systemic lupus erythematosus (SLE) and systemic sclerosis (SSc), separately. It is expected to obtain IND approval from the NMPA for B-cell malignancies in 2026. Phase Ib registration study is planned to be initiated in 2026 in
In addition, multiple products against different targets are currently under development: KJ-C2320 against CD38 for acute myeloid leukemia (AML); KJ-C2526 against NKG2DL for AML, other malignancies and senescence; CT1390B against CLL1 for AML; KJ-C2527 against Claudin18.2 for gastric cancer.
On
In vivo CAR-T product candidates in development
Apart from CAR T-cell products manufactured via in vitro gene editing, CARsgen is also developing in vivo CAR T-cell products. CARsgen's proprietary lentiviral-based CARvivo™ platform demonstrates excellent T cell transduction and targeting specificity. KJ-C2529 is an in vivo CAR T-cell product candidate against CD19/CD20 deploying our CARvivo™ platform for the treatment of B-cell lymphoma. An IIT is expected to be initiated in 2026 for the treatment of R/R B-NHL.
Expand CAR-T commercial manufacturing base
CARsgen is actively preparing for capacity expansion, enhancing the manufacturing capabilities for CAR T-cell therapies that meet international standards to support the commercialization of multiple products and strengthen its global competitiveness. On
About
CARsgen is a biopharmaceutical company focusing on developing innovative CAR T-cell therapies to address the unmet clinical needs including but not limited to hematologic malignancies, solid tumors and autoimmune diseases. CARsgen has established end-to-end capabilities for CAR T-cell research and development covering target discovery, preclinical research, product clinical development, and commercial-scale production. CARsgen has developed novel in-house technologies and a product pipeline with global rights to address challenges faced by existing CAR T-cell therapies. Efforts include improving safety profile, enhancing the efficacy in treating solid tumors, and reducing treatment costs, etc. CARsgen's mission is to be a global biopharmaceutical leader that provides innovative and differentiated cell therapies for patients worldwide and makes cancer and other diseases curable.
Forward-looking Statements
All statements in this press release that are not historical fact or that do not relate to present facts or current conditions are forward-looking statements. Such forward-looking statements express the Group's current views, projections, beliefs and expectations with respect to future events as of the date of this press release. Such forward-looking statements are based on a number of assumptions and factors beyond the Group's control. As a result, they are subject to significant risks and uncertainties, and actual events or results may differ materially from these forward-looking statements and the forward-looking events discussed in this press release might not occur. Such risks and uncertainties include, but are not limited to, those detailed under the heading "Principal Risks and Uncertainties" in our most recent annual report and interim report and other announcements and reports made available on our corporate website, https://www.carsgen.com. No representation or warranty is given as to the achievement or reasonableness of, and no reliance should be placed on, any projections, targets, estimates or forecasts contained in this press release.
For more information, please visit https://www.carsgen.com/
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/carsgen-therapeutics-announces-2025-annual-results-302706644.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/carsgen-therapeutics-announces-2025-annual-results-302706644.html
SOURCE